You’ve been told the same thing a million times: The best way to prevent the spread of coronavirus is to wash your hands.
Brookhaven National Lab
It’s true. But why?
It’s because soap — regular soap, fancy honeysuckle soap, artisan soap, just any soap — absolutely annihilates viruses. It has to do with how the soap molecules interact with the virus.
Soap is made up of two-sided molecules. One side is attracted to water; the other side is attracted to fat. And viruses are made up of material surrounded by a coating of proteins and fat. When viruses interact with soap, that fat coating gets ripped out by the soap molecules. Soap literally demolishes viruses.
Of course, it takes time for this effect to happen: 20 seconds, to be specific.
Watch the video above to learn how this process works or READ THE FOLLOWING FROM
senior science reporter Brian Resnick.
As Covid-19 cases in the US surge to more than 9000 and fear sweeps the country, there’s one consumer product critical to our great national battle to “flatten the curve,” or slow the epidemic: soap. Humble, ancient, cheap, effective soap.
Respiratory viruses — like the novel coronavirus, the flu, and the common cold — can be spread via our hands. If someone is sick, a hand can touch some mucus and viral particles will stick to the hand. If someone is well, hands act like sticky traps for viruses. We can pick up droplets that contain the virus, and they’ll stay on our hands, and perhaps enter our bodies if we touch our hands to our faces.
That’s why our hands are the front lines in the war against Covid-19. The Centers for Disease Control and Prevention (CDC) recommends washing hands with soap and water as the top way to clean our hands. “But if soap and water are not available, using a hand sanitizer with at least 60% alcohol can help,” the CDC says.
The CDC prioritizes soap. Yet, per news reports, people have been stocking up and hoarding sanitizer. The sanitizer situation is growing absurd: The Atlantic reported on a man who sold a bottle of Purell on eBay for $138. Hand sanitizer containing over 60 percent alcohol works against Covid-19 and is a good option when you’re not near a sink. But it’s getting harder to find than a hypodermic needle in a haystack.
Sanitizer might feel like a modern-day, scientific, and more clinical upgrade to soap. But I’m here to tell you that soap — all sorts of it: liquid, solid, honeysuckle-scented, the versions inexplicably only marketed to men or women — is a badass, and even more routinely effective than hand sanitizer. We should be excited to use it, as much as possible.
That’s because when you wash your hands with soap and water, you’re not just wiping viruses off your hands and sending them down the drain. You’re actually annihilating the viruses, rendering them harmless. Soap “is almost like a demolition team breaking down a building and taking all the bricks away,” says Palli Thordarson, a chemistry professor at the University of New South Wales, who posted a viral Twitter thread on the wonders of soap.
In a recent phone call, he explained why soap is such an effective Covid-19 killer and why it’s so important to soap your hands for at least 20 seconds.
First up: What is soap?
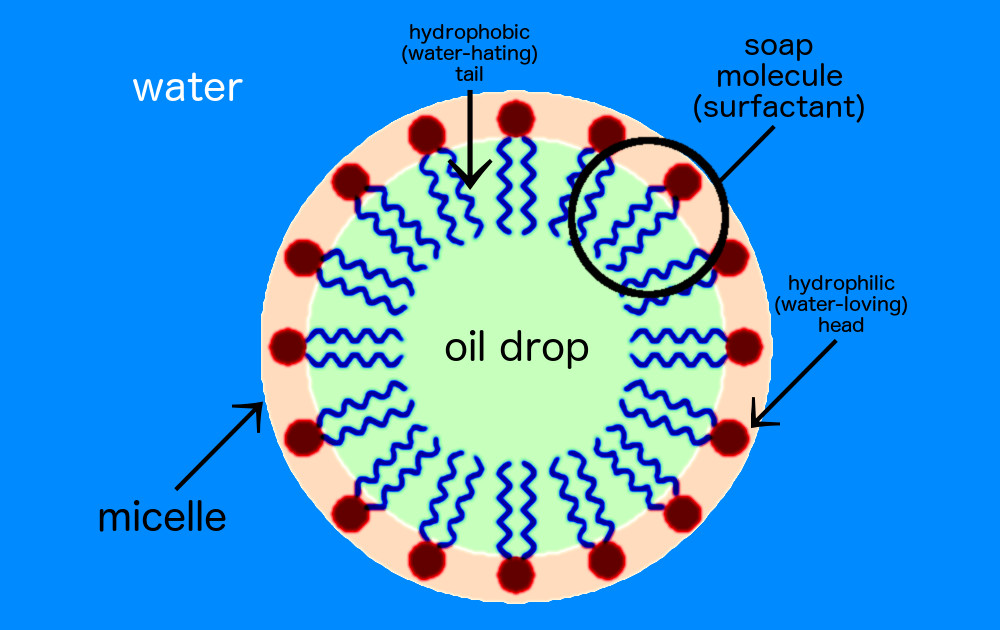
Soap, Thordarson explains, is common phrase for what chemists call “amphiphiles.” These are molecules that have a dual nature. One end of the molecule is attracted to water and repelled by fats and proteins. The other side of the molecule is attracted to fats and is repelled by water. (If you’re looking out for product labels, the most common soap is “sodium laureth sulfate” — it’s a detergent that’s often mixed with other chemicals to both clean our hands and not damage our skin.)
It’s this dual-nature chemical construction that makes soap so effective. “When you buy a conventional soap, it consists of a mixture of these amphiphiles,” Thordarson explains. And they all do the same thing.
Think about what happens when you pour some olive oil into water. The oil pools up in a mass that floats. “That’s because fats don’t mix with water,” he says. But mix some soap into the oil and water and the oil will disperse. Basically, that happens because the soap is attracted to the grease, via its fat-loving side, but then tears it up, pulling it into the water via its water-loving side. It’s a one-two punch. Surround the oil particles and move them away from one another.
Now, lucky for us, coronaviruses are a bit like the oil mentioned in the above example: bits of genetic information — encoded by RNA — surrounded by a coat of fat and protein. Thordarson likes to call viruses “nano-sized grease balls.” And grease balls, no matter the size, are the exact type of thing soap loves to annihilate.
1/25 Part 1 – Why does soap work so well on the SARS-CoV-2, the coronavirus and indeed most viruses? Because it is a self-assembled nanoparticle in which the weakest link is the lipid (fatty) bilayer. A two part thread about soap, viruses and supramolecular chemistry #COVID19 pic.twitter.com/OCwqPjO5Ht
— Palli Thordarson (@PalliThordarson) March 8, 2020
How soap destroys viruses
The soap takes care of the virus much like it takes care of the oil in the water. “It’s almost like a crowbar; it starts to pull all the things apart,” Thordarson says.
One side of the soap molecule (the one that’s attracted to fat and repelled by water) buries its way into the virus’s fat and protein shell. Fortunately, the chemical bonds holding the virus together aren’t very strong, so this intrusion is enough to break the virus’s coat. “You pull the virus apart, you make it soluble in water, and it disintegrates,” he says.
Then the harmless shards of virus get flushed down the drain. And even if it the soap doesn’t destroy every virus, you’ll still rid them from your hands with soap and water, as well as any grease or dirt they may be clinging to. Soap will also wash away bacteria and other viruses that may be a bit tougher than coronavirus, and harder to disintegrate.
The trick is this all takes a little time to happen, and that’s why you need to take at least 20 seconds to wash your hands.
First off, your skin is wrinkly, and it takes time for soap to penetrate into all the tiny folds and demolish the viruses that lurk within. Then the soap needs a few moments to do its chemical work. “You do need a bit of time for all the soap to interact back and forth with the virus particle,” he says. Twenty seconds should do the trick just fine.
Alcohol, the main ingredient in hand sanitizer, can destroy viruses, too. Sanitizers “actually work in a similar way, the alcohol molecules are somewhat amphiphiles,” he says. The thing is, you need a very high concentration of alcohol to achieve the same effect. (Chemicals called quaternary ammonium compounds — the main ingredient in Lysol — kill viruses too but can be a bit harsher on the skin.)
The CDC recommends a sanitizer that’s 60 percent alcohol, so beware of sanitizers or wipes on the market that don’t meet this standard (or contain alcohol at all). Hand sanitizer is useful, but it can fail in un-ideal situations. If your hands are wet or sweaty when you use the sanitizer, that can dilute it and diminish its effectiveness. Also, sanitizer doesn’t clean your hands of sticky grease to which viruses can also adhere.
“Soap doesn’t really fail easily,” Thordarson says. It doesn’t really matter the formulation of soap, either. You don’t need “antibacterial soap” — which the Food and Drug Administration advises to skip altogether due to a lack of evidence of its usefulness. And you don’t need a super-harsh detergent like you’d put in your dishwasher or laundry machine. Simple soap works fine. “As long as you give it a little bit of time, it will do its job.”
All of this, at least, makes me excited to wash my hand more. As I’m washing with soap and water and counting to 20, I’m going to imagine a battle being waged on the nano-scale in the teeny-tiny folds of my skin. The soap is charging in, sticking to viruses (as well as dirt and other grease), and tearing them apart in brutal, heroic fashion. It’s almost like the Avengers, but better. Because it’s real. And it can help stop the spread of this outbreak.
VOX

Leave a Reply
You must be logged in to post a comment.